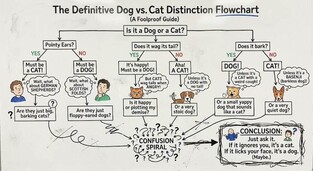
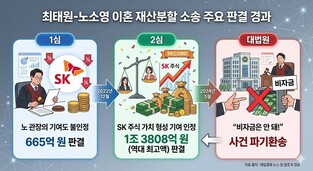
 |
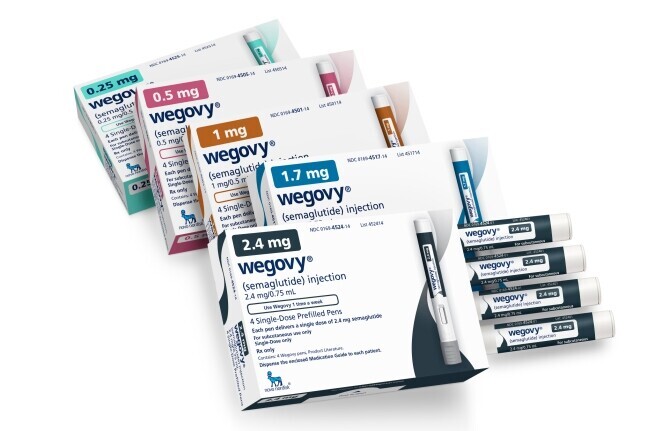
Photo courtesy of Wegovy |
[Alpha Biz= Paul Lee] Novo Nordisk’s obesity drug Wegovy (semaglutide) has received U.S. Food and Drug Administration (FDA) approval in an oral pill form, officially opening the market for oral obesity treatments and raising expectations for R&D progress among Korean developers such as D&D Pharmatech, Unovia, and Celltrion.
On December 22 (local time), Novo Nordisk announced that Rybelsus (oral semaglutide 25 mg), taken once daily, was approved for weight loss and long-term weight maintenance in overweight and obese adults, as well as for reducing the risk of major adverse cardiovascular events (MACE). The drug is scheduled to launch in the U.S. in January next year and will become the world’s first oral GLP-1–based obesity treatment.
In a Phase 3 clinical trial involving 307 overweight or obese adults without diabetes, Rybelsus demonstrated an average weight reduction of 16.6%. Novo Nordisk set the U.S. list price at USD 149 per month.
알파경제 Paul Lee 특파원(hoondork1977@alphabiz.co.kr)